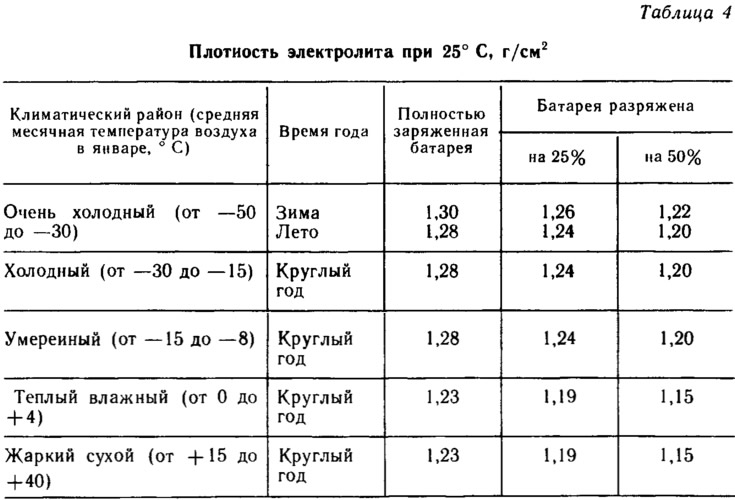
- Rated voltage, V - 12
- Rated capacity at a 20-hour discharge mode and an electrolyte temperature of 25°C at the beginning of the discharge, Ah - 55
- Discharge current at 20-hour discharge mode, A - 2.75
- Discharge current in the starter discharge mode and electrolyte temperature -18°C, A - 255
On cars VAZ-2108 is installed «underserved» rechargeable battery type 6ST-55A. It is designed to start the engine and power consumers when the engine is not running, when the generator does not generate electricity.
Building 1 (pic. 96) made of translucent plastic - polypropylene and divided by impenetrable partitions into sections, in which six batteries connected in series with a voltage of 2 V each are placed. Cover 2, common to the entire body, is also made of polypropylene and welded to the body using contact heat welding. The lid has six openings for filling electrolyte and controlling its level. Holes are closed with plugs 6.
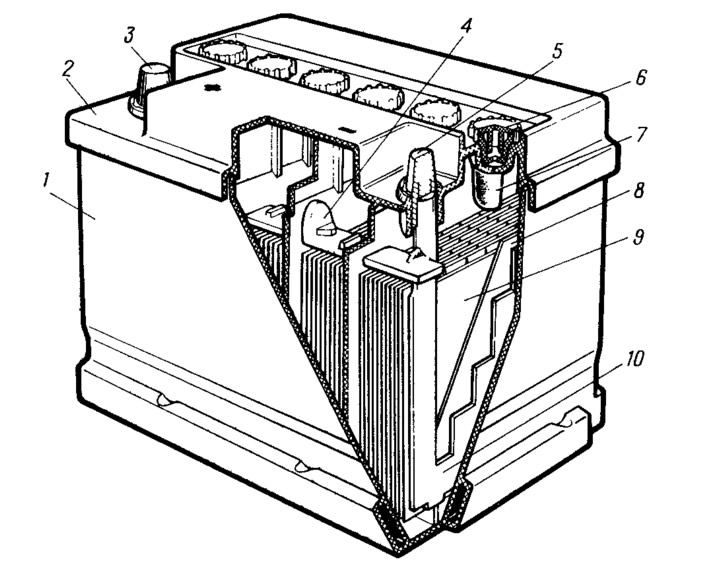
Pic. 96. Battery:
1 - body; 2 - cover; 3 positive conclusion; 4 - interelement connection; 5 - negative conclusion; 6 - cork; 7 - indicator for checking the electrolyte level; 8 - separator; 9, 10 - positive and negative plates.
Each battery contains a block of alternately arranged positive 9 and negative 10 plates. They are made in the form of a grid filled with a porous active mass of lead and lead oxides. Plates of the same polarity are assembled into semi-blocks and welded to common plates - barettes.
Lattice plates cast from lead alloy (with added calcium) with low antimony content. This slows down the process of self-discharge of the battery and reduces the decomposition of the electrolyte at the end of the charge. As a result, the complexity of battery maintenance has decreased, since it has become possible to check the level and density of the electrolyte less often. Therefore, these batteries are called «under-served» or even «unattended».
Separators 8 made of thin microporous polyvinyl chloride are installed between the plates in the blocks. They serve to isolate dissimilar plates from each other, as well as to eliminate the vibration of the plates during shaking. The small thickness and large porosity of the separators reduces the internal resistance of the battery and makes it possible to obtain a large discharge current. And this makes it easier to start the engine at sub-zero electrolyte temperatures.
The electrolyte in the battery is a solution of sulfuric acid in distilled water. When the battery is discharged, the sulfuric acid of the electrolyte interacts with the active mass of the plates and turns it into lead sulfate (white color), while the amount of acid in the electrolyte decreases and its density decreases.
When the battery is charged under the action of the current passing through the battery, the reverse process occurs. Lead sulfate in the active mass of the positive plates is converted to lead peroxide (Brown), and at the negative plates it turns into spongy lead (gray color), while sulfuric acid is released into the electrolyte and its density increases. At the factory, cars are equipped with batteries with an electrolyte density of 1.26... 1.28 g / cm3 (for the tropics - with a density of 1.22... 1.24 g / cm3). Depending on the climatic region in which the car is operated, the density of the electrolyte must correspond to that indicated in Table. 4.